ProcessUnity Industries
ProcessUnity for Life Sciences
Ensure regulatory compliance and make data-driven decisions.

The Life Sciences industry uses third parties for several reasons, including access to expertise and specialization, cost efficiency, flexible capacity, risk sharing, and speed-to-market. Hence third-party risk management is essential for maintaining product quality, complying with regulations, protecting sensitive information, ensuring business continuity, and upholding ethical and legal standards.
Life Sciences companies need a comprehensive risk management solution such as ProcessUnity Third-Party Risk Management and ProcessUnity Cybersecurity Risk Management to automate and streamline risk assessment, management and mitigation through the entire supplier relationship life cycle.
Proactively Mitigate Risk and Ensure Regulatory Compliance
Use our platform to score inherent risk and identify vendors with a high exposure to risk. Customize vendor due diligence according to their risk profile. Employ assessment questionnaires, content feeds from data providers, internal assessments, and more to test relevant controls, identify gaps and mitigate risks. Ensure suppliers comply with Good Manufacturing Practices (GMP), regulatory approval processes and standards set by FDA and EMA, EHS and anti-corruption regulations, and others.
Protect Data and Assets
Identify vendors with access to NPI/PII (non-public information/personally identifiable information) to safeguard against data privacy threats. Knowing which vendors handle your sensitive data lets you focus your assessments and ensure compliance with relevant regulations and service level agreements.
Reduce Cybersecurity Risk
Analyze extensive third-party data to spot potential vulnerabilities and threats to your infrastructure from your partners. ProcessUnity streamlines analysis, ensuring no potential cyber risks from partners are overlooked.
Drive Consistency and Accountability with an Automated Workflows
Evaluate third parties using tailored questions mapped to regulations, standards, and enterprise controls. Customize questionnaires based on each vendor’s service type and inherent risk profile for high-quality responses. Document issues from due diligence, continuous monitoring, and KPI tracking, then connect them to controls to develop and track effective mitigation actions.
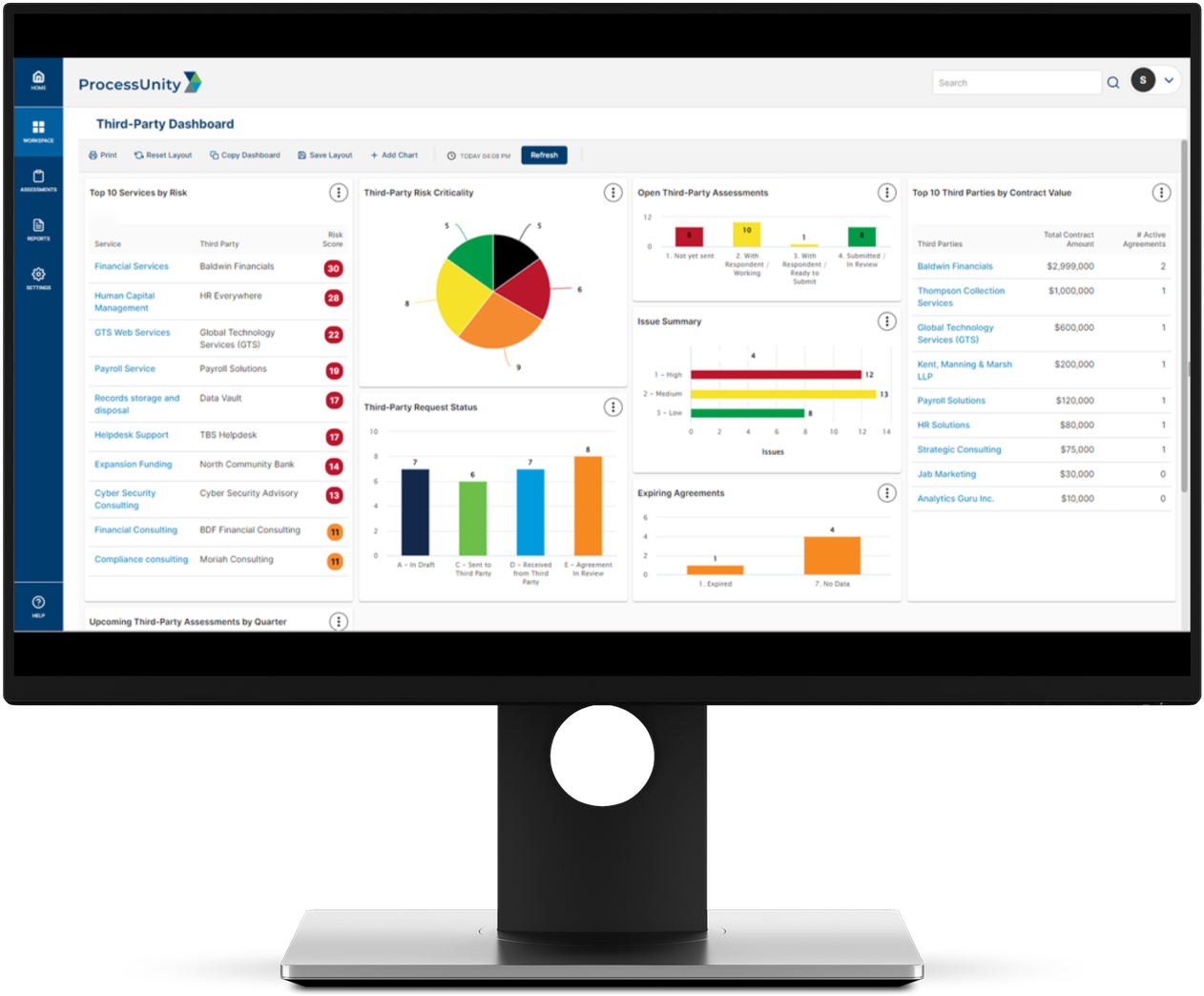
Increase Visibility into Risk and Performance for Key Business Stakeholders
Utilize our platform to get access to real-time reporting and configurable dashboards. Gain an immediate understanding of your third-party risk posture, key performance metrics, and status of any request.
Request a demo below to learn more about ProcessUnity for Life Sciences.
Request a Demo: ProcessUnity's Risk Management Platform
Schedule your personalized demo of our award-winning software and start your journey to a more mature, automated program.
Request a Demo: ProcessUnity's Risk Management Platform